 The horizontal rows of the periodic table are known as the Periods.The shortest groups of the periodic table are from transition metals groups and the longest is Group 3.Alkaline Earth Metals Similar to the group 1 elements, these elements change upon interacting with. Alkali Metals All the elements in group 1 are regarded as Alkali Metals, mainly because they have a chemical reaction with water, which will result in alkali solutions. Groups are distinguished into seven different sections: Alkali metals (Group 1), Alkaline earth metals (Group 2), Transition metals (Group 3-12), Pnictogens (Group 15), Chalcogens (Group 16), Halogens (Group 17), and Noble gases (Group 18). Currently, in the modern Periodic Table, there are 8 groups, such as.The outermost electrons of all elements in the same group have the same chemical behaviour.A period on the periodic table is a row of chemical elements.All elements in a row have the same number of electron shells.Each next element in a period has one more proton and is less metallic than its predecessor. Most periodic tables provide additional data (such as atomic mass) in a box that contains each element’s symbol. In the periodic table of the elements, each numbered row is a period. A modern version is shown in Figure 2.6.1 2.6. :max_bytes(150000):strip_icc()/periodic-table-165930186-590f2d703df78c92832fe141.jpg)
The groups are differentiated on the basis of the number of valence electrons and the level of reactivity. The periodic table is one of the cornerstones of chemistry because it organizes all the known elements on the basis of their chemical properties. 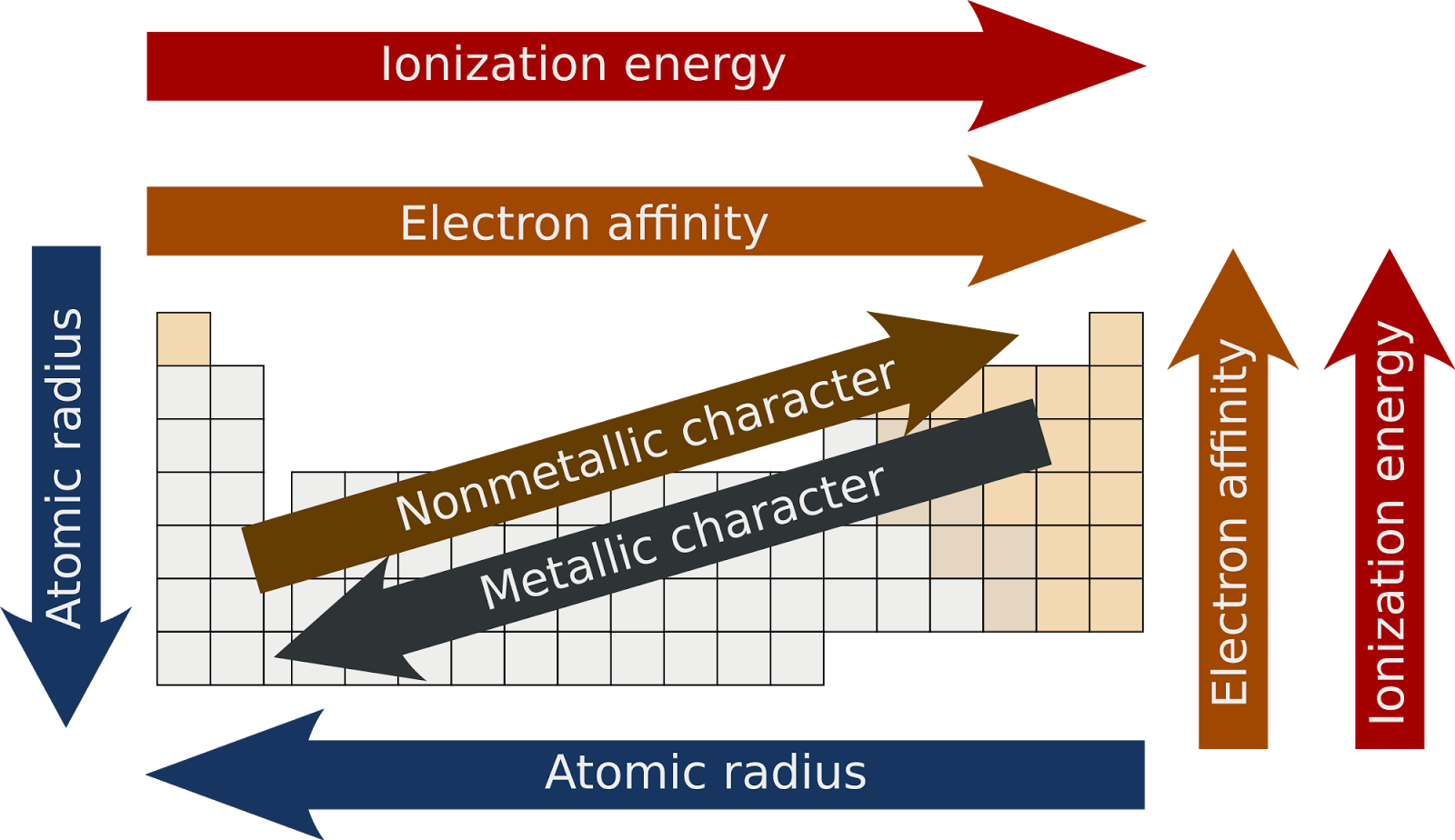
There is a total of 18 groups in the periodic table.Other people consider the transition metals to include any d -block element on the periodic table. A transition metal is an element with a partially-filled d subshell or the capacity to produce cations with an incomplete d subshell. There are mainly two sections to the periodic table: Groups and Periods The most common definition of a transition metal is the one accepted by the IUPAC. In older IUPAC group numbering systems, the main group elements are groups IA, IIA, and IIIA to VIIIA. These are elements in group 1 and group 2 (s-block) and groups 13 through 18 (p-block). JEE Main 2022 Question Paper Live Discussion The main group elements are the chemical elements belonging to the s-block and p-block on the periodic table.Difference Between Selling And Marketing.TS Grewal Solutions Class 11 Accountancy.TS Grewal Solutions Class 12 Accountancy.CBSE Previous Year Question Papers Class 12.CBSE Previous Year Question Papers Class 10.NCERT Solutions For Class 6 Social Science.NCERT Solutions for Class 7 Social Science.NCERT Solutions for Class 8 Social Science.They will be responsible for presenting the information orally to the class. They will complete a fact sheet, a poster, and write a jingle. Students will adopt an element to research. Reading the Periodic Table (periods, groups).Define and name examples of substituted hydrocarbons.Define hydrocarbons and describe the kinds of carbon chains in them.Identify the properties many organic compounds have in common.Explain how carbon can form bonds in many different arrangements.Compare the physical and chemical properties of nonmetals with those of metals and describe the properties of metalloids.Locate nonmetals and metalloids in the periodic table.Identify and describe different groups of metals and describe how the reactivity of metals changes across the periodic table. 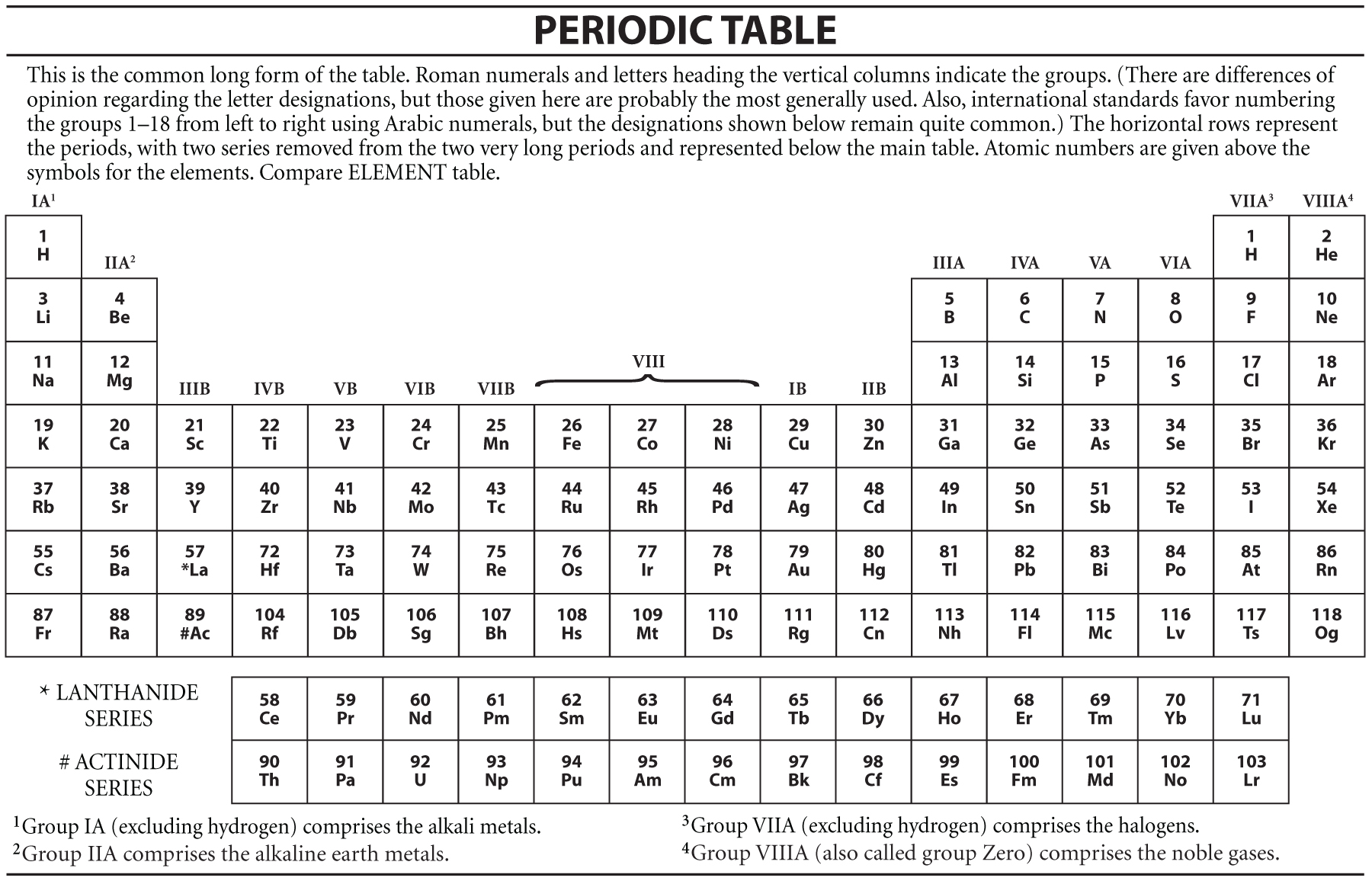
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
